by Murota & Saito
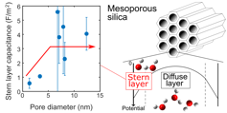
Water in confinement becomes more structured than bulk water, and its properties, such as the dielectric constant, change. It remains unclear, however, how the interfacial reactions in confinement, such as the adsorption of ions on the surfaces of small pores, differ from those in larger spaces. We focused on the deprotonation reaction of hydroxyl groups, a fundamental surface reaction, and investigated the dependence of the surface charge density on pore size by determining the surface charge densities of six types of mesoporous silicas with micropores and mesopores at different ionic strengths and pH levels from batch titration tests. The surface complexation model assuming a potential distribution based on the Poisson-Boltzmann equation in cylindrical coordinates was fitted to the obtained surface charge densities to relate the electrostatics near the surface to the surface reaction. The results showed that the absolute values of the surface charge densities decreased with decreasing pore diameter due to the overlap of the electrical double layers. Furthermore, the capacitance of the Stern layer optimized by fitting decreased with decreasing pore diameter, especially in pores smaller than 4 nm in diameter, which suggested that the dielectric constants of water decreased near the surfaces of small pores.